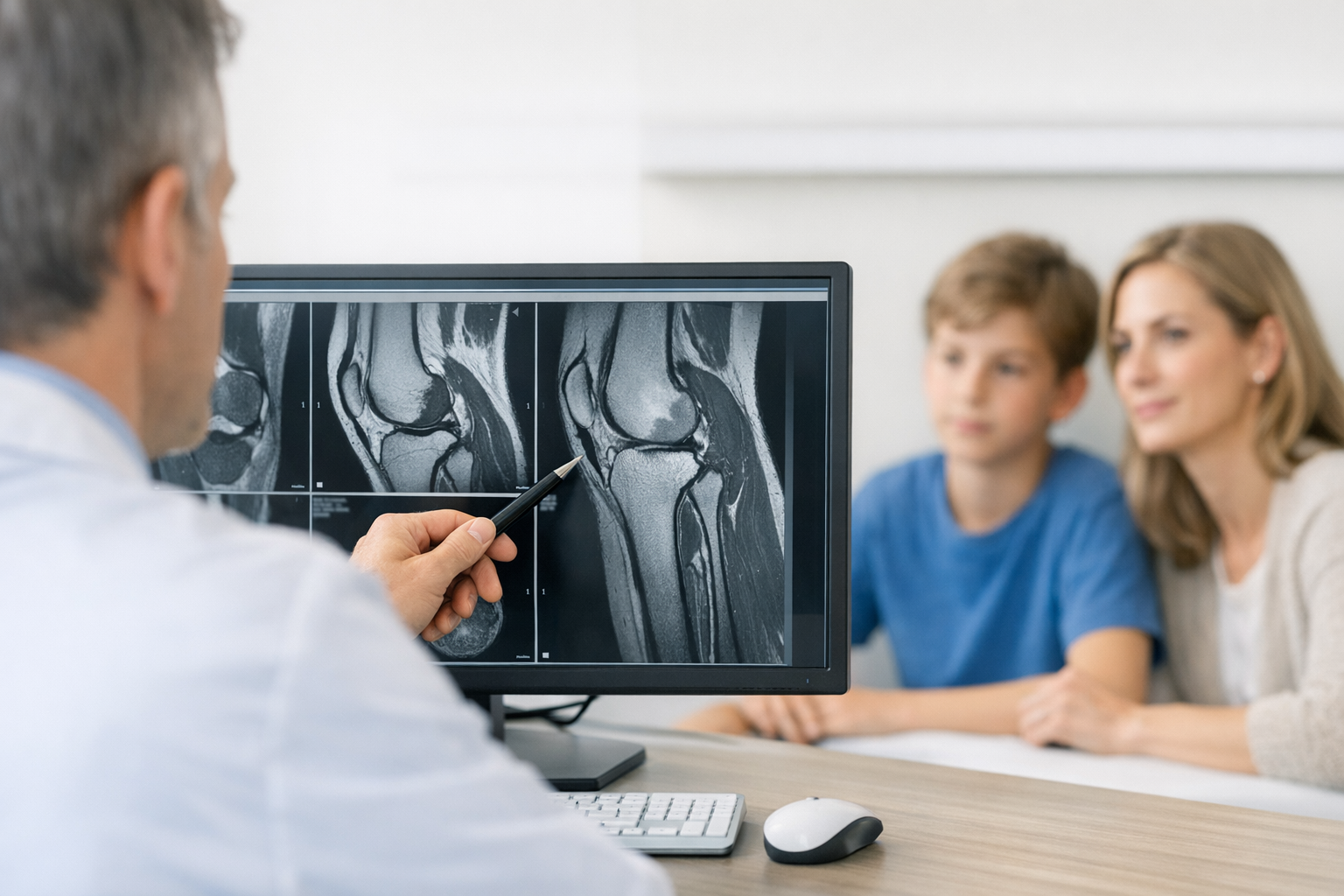
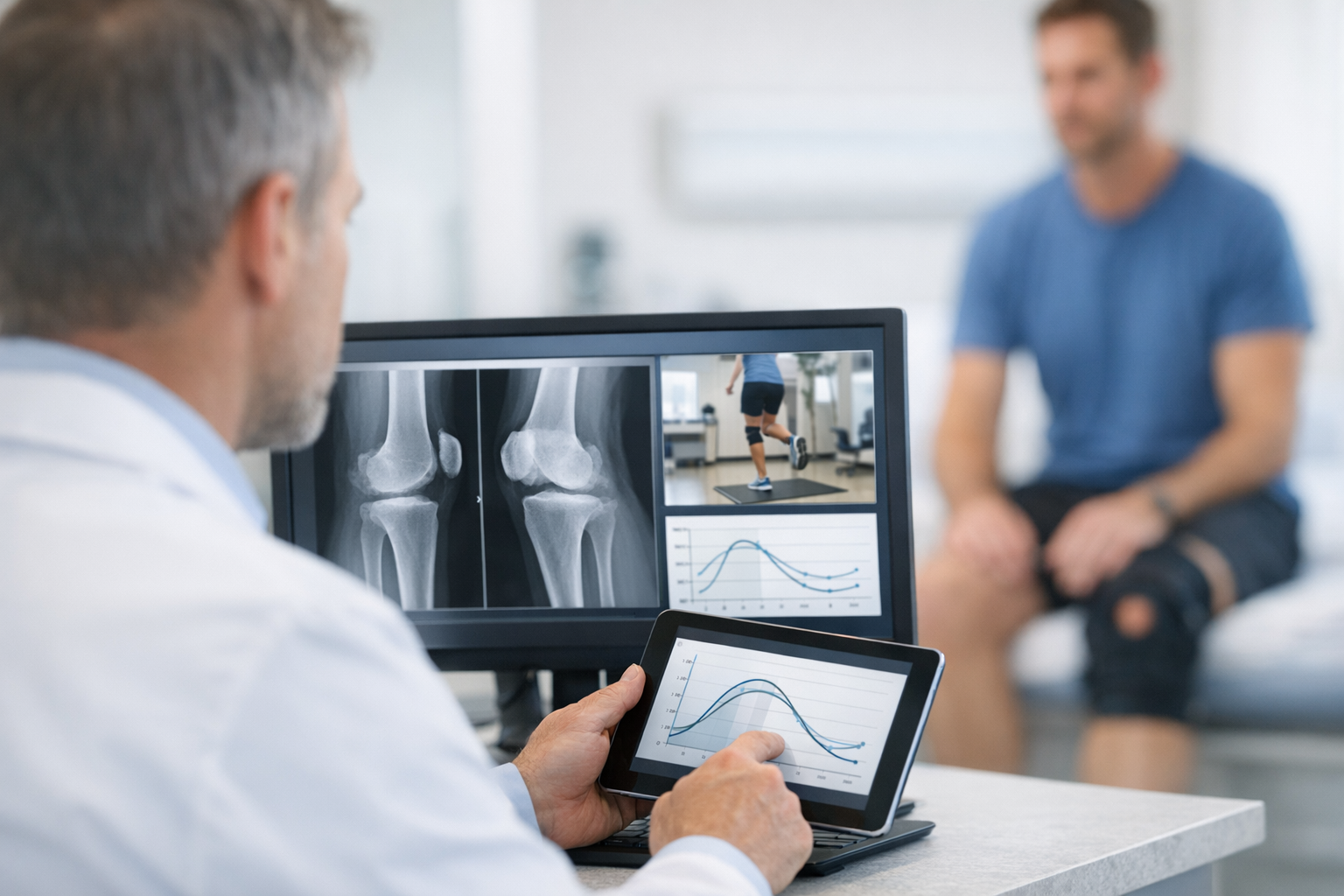
In knee ligament care, the practical challenge is rarely “is something torn?” but “how unstable is this knee in function, in more than one plane, and how does that change with treatment?” dynamic knee laxity testing is a workflow tool for answering that question with repeatable, side-to-side comparisons that can be tracked across time. It sits beside the history, clinical exam, and imaging, adding quantifiable information about multi-axis knee stability that is hard to capture with manual grading alone, especially when swelling, guarding, or examiner variability affects the exam. Used thoughtfully, it can improve triage, clarify borderline cases, and support shared decisions around rehab progression and return-to-sport, while remaining complementary to MRI and clinician-led diagnosis.
1) Intake to first-contact triage: set up the instability question
Start with a triage mindset: identify red flags, define likely injury patterns, and decide whether immediate immobilization, imaging, or referral is needed. A structured pathway such as an urgent-care knee ligament injury triage algorithm helps standardize what happens in the first 24 to 72 hours when pain and effusion can distort exam findings.
1.1) Key intake fields that influence the testing plan
- Mechanism: non-contact pivot, valgus load, hyperextension, dashboard injury.
- Timing and swelling: hemarthrosis within hours raises suspicion for ACL rupture or osteochondral injury.
- Symptoms: giving way, shifting, inability to decelerate, apprehension on cutting.
- Baseline risk: prior ACL reconstruction, generalized laxity, high-demand athlete, contact sport.
- Contraindications to stress testing today: suspected fracture, gross dislocation, neurovascular symptoms, locked knee suggesting bucket-handle tear.
From an operational perspective, the goal at this stage is to define the “instability question” you need to answer later. In many clinics, dynamic knee laxity testing is scheduled after acute pain settles enough to permit consistent positioning and repeatable loads, rather than forcing a low-quality baseline on day one.
1.2) Early decision: “likely isolated ACL” vs “multiligament or corner involvement”
Use simple pattern recognition to decide whether you can proceed with standard instability quantification, or whether you should prioritize protective management and specialist assessment:
- Suspect MCL involvement with valgus mechanism, medial tenderness, and gapping at 30 degrees (and potentially at 0 degrees if more extensive).
- Suspect LCL/PLC involvement with varus/hyperextension mechanism, posterolateral pain, or external rotation recurvatum pattern.
- Suspect PCL with posterior sag, posterior drawer, or dashboard mechanism.
This stratification matters because multi-planar and rotational laxity often reflects combined injuries, and your subsequent measurement plan should mirror the suspected planes of instability.
2) Baseline exam plus dynamic knee laxity testing: standardize the “time zero” dataset
Before you quantify anything, standardize how you examine. In busy clinics, variation in knee flexion angle, tibial rotation, and patient relaxation is a major source of noise. A consistent template such as objective measures in the orthopaedic knee examination helps align the team on what “good quality data” looks like.
2.1) Build the core dataset (what to record every time)
Pair a traditional clinical exam with a minimal set of repeatable metrics:
- Effusion (stroke test or sweep) and pain level at rest and with testing.
- ROM (especially extension deficit that may influence pivot shift and rotational maneuvers).
- Manual tests with graded notes: Lachman (endpoint quality), anterior drawer, pivot shift (if tolerated), valgus stress for MCL, varus stress for LCL, posterior drawer for PCL, dial test when indicated.
- objective knee laxity measurement outputs: ensure the same knee angle, stabilization strategy, and patient instructions each visit.
Operationally, it helps to treat measurement as a “protocol” rather than a device reading. For a concise overview of outputs and common approaches, see knee laxity testing methods and outputs.
2.2) Interpretation anchor: side-to-side difference first, then absolute values
In ACL pathways, the most actionable frame is often the side-to-side difference knee laxity compared with the contralateral limb, interpreted alongside symptoms and manual endpoint quality. This is where dynamic knee laxity testing can be most clinically useful: it can reduce reliance on “feel” when guarding or examiner experience limits confidence.
Pitfalls to avoid:
- Testing through significant pain or spasm (false low laxity).
- Inconsistent tibial rotation control between visits (apparent “change” that is positioning, not biology).
- Using a single plane metric to overrule a strong clinical pattern of multi-ligament injury.
2.3) Operational checklist (who does what)
- Clinician: determines suspected structures and acceptable maneuvers that day.
- Physio/ATC: ensures relaxation cues, consistent positioning, documents symptoms during testing.
- Research coordinator (if applicable): confirms protocol adherence, stores raw curves, logs covariates (effusion, time post injury, analgesia).
If your clinic uses dynamic protocols, align your steps with established concepts and repeatability principles outlined in dynamic knee testing concepts and protocols. That consistency is what allows dynamic knee laxity testing to function as a longitudinal metric rather than a one-off number.
3) Multi-axis and rotational workflow: connect translation, valgus, and pivot mechanics
The knee rarely fails in a single plane during sport. For ACL injury, rotational components (anterolateral structures, meniscus, and neuromuscular control) can dominate symptoms even when anterior translation looks “borderline.” A workflow that explicitly maps measures to planes is how you turn numbers into decisions.
3.1) Map tests to the suspected structure set
- ACL-dominant: Lachman and anterior translation metrics, plus rotational assessment if giving-way is the main symptom.
- ACL + MCL: add valgus laxity quantification and document medial pain and endpoint; consider that MCL laxity can alter perceived pivot behavior.
- ACL + PLC/LCL: prioritize rotational and varus-plane assessment and avoid over-simplifying to “ACL laxity only.”
In rotational pathways, pivot shift quantification can be considered when the patient can tolerate it and the team can standardize tibial rotation and starting position. The pivot shift remains partly examiner dependent, so any quantification approach should be integrated cautiously and interpreted with the broader clinical picture.
3.2) Where MRI fits (and why it stays complementary)
MRI is typically needed to assess associated injuries (meniscus, cartilage, bone bruise patterns) and to support pre-operative planning when reconstruction is being considered. The value of dynamic knee laxity testing is different: it can add functional information about instability magnitude and side-to-side differences that imaging alone may not fully reflect, especially in suspected partial ACL tears or equivocal imaging.
For an operational pathway that deliberately combines these inputs, see the workflow combining MRI with instability testing for borderline cases. Use it to standardize how you escalate from clinical suspicion to imaging, and when you revisit instability metrics after swelling and pain improve.
When surgeons consider adding an anterolateral procedure for rotational control, it is helpful to remember that technique choices and indications vary across centers. Outcomes research such as Andre et al. (2026) can inform discussions around combined ACL and ALL reconstruction strategies, but decisions remain individualized and context specific.
4) Instrumented arthrometers in a multi-axis pathway (clinic and research integration)
This section operationalizes how to embed instrumented measures without letting the tool drive the diagnosis. Done well, an instrumented arthrometer knee workflow produces repeatable curves and side-to-side comparisons that complement manual grading and MRI findings.

4.1) What the evidence supports you doing operationally
Two practical evidence points relate to reliability and feasibility:
- Feasibility and reliability of a robotic approach has been examined in Nascimento et al. (2024), supporting the idea that standardized setup and protocol adherence are central to repeatable outputs in both healthy and ACL injured or reconstructed populations.
- Reliability and the learning curve for another commonly used system were evaluated in Unal et al. (2024), reinforcing that team training and consistent positioning can materially influence the repeatability of measurements.
Those themes align with day-to-day implementation lessons: schedule onboarding sessions, maintain a single clinic protocol, and log covariates (time since injury, effusion, analgesia, brace use) so changes in curves are interpretable.
For device-specific context on reliability, see the Dyneelax reliability for knee ligament assessment. For a clinical example of how anterior laxity can vary with concomitant pathology, review the report on GNRB findings in ACL-deficient knees with lateral meniscal tears.
4.2) Where GNRB and Dyneelax fit (without overclaiming)
In practice, clinics that use instrumented systems may incorporate GNRB knee laxity measures or a Dyneelax arthrometer protocol to standardize anterior translation across visits, while still interpreting results in the context of clinical exam and MRI. If you want device overviews, see GNRB arthrometer ACL assessment and Dyneelax knee arthrometer.
Important positioning: these measurements do not replace MRI. They can complement MRI by quantifying functional side-to-side instability and helping clarify equivocal or borderline presentations, while MRI remains valuable for defining meniscal, chondral, and osseous associated injuries.
4.3) Add-on variables that change how you interpret laxity
Interpretation improves when you treat laxity as biology plus context:
- Meniscal status and bony morphology can influence anterior laxity in ACL-deficient knees, as explored in Huang et al. (2025).
- Graft tissue maturation is not just a timeline issue; early graft composition and imaging markers may relate to measured laxity, as examined in Figueroa et al. (2025).
Operational takeaway: if the curve changes unexpectedly, do not assume “failure.” Check effusion, pain inhibition, measurement setup, and new meniscal symptoms, then consider whether MRI is needed to evaluate new intra-articular pathology.
At this stage, dynamic knee laxity testing becomes a repeatable “functional biomarker” rather than a one-time diagnostic add-on, especially when you standardize who positions the knee and how you document patient response.
5) Longitudinal monitoring: linking stability metrics to rehab and surgical decisions
The most valuable workflows treat measurements as serial data points that support decisions, not as pass-fail gates. In ACL pathways, ACL graft laxity monitoring is typically most informative when paired with symptoms, strength, hop performance, movement quality, and psychological readiness.
5.1) Choose timepoints that align with clinical decision nodes
Instead of measuring “because we can,” tie dynamic knee laxity testing to moments when you might change management:
- Post-acute baseline once swelling and guarding allow repeatable positioning.
- Prehabilitation checkpoint (especially if surgery is being considered).
- Early post-op when the protocol allows safe testing (clinic-specific), focusing on trend and side-to-side comparisons rather than single numbers.
- Late rehab and return-to-sport decision window.
To operationalize those checkpoints, connect your measurements to rehab milestones versus knee stability metrics. This helps the team avoid measuring too early (noisy data) or too late (missed opportunity to adjust rehab).
5.2) How to interpret change over time (decision aid)
Use a simple, clinician-led decision aid when serial results shift:
- Confirm data quality: same protocol, same knee angle, similar symptoms and effusion?
- Check the patient story: any new giving-way episode, new joint-line pain, or swelling spike?
- Reconcile planes: does increased translation come with rotational symptoms or new valgus laxity suggesting MCL involvement?
- Escalate appropriately: consider repeat clinical exam and, when indicated, MRI for associated injury assessment and planning.
- Adjust the plan: load management, neuromuscular focus, bracing, or surgical discussion based on the full picture.
This is where dynamic knee laxity testing can prevent overreaction to a single metric by pushing the team to verify repeatability before acting.
5.3) Go beyond millimeters when helpful: compliance and stiffness concepts
Some teams find that focusing only on displacement misses clinically relevant mechanical behavior. Concepts like compliance and stiffness can add nuance to graft and tissue behavior, and they may help explain why two patients with similar translation values feel different functionally. For a practical overview, review why compliance and stiffness metrics can outperform simple laxity.
In late rehab, if objective stability is being used alongside functional tests, integrate it with a structured approach such as return-to-sport criteria using objective stability. In that context, dynamic knee laxity testing can act as a complementary stability signal, not a standalone clearance tool.
6) Closing: key takeaways and next steps
Key takeaways:
- dynamic knee laxity testing is most useful when it is protocolized, side-to-side, and repeated at decision-making timepoints.
- Multi-axis thinking matters: anterior translation, valgus laxity (MCL), and rotational instability should be mapped to suspected structures and symptoms.
- Arthrometer outputs can complement clinical exam and MRI, especially in borderline functional instability, but do not replace imaging when associated injuries or surgical planning are in scope.
Next steps for implementation:
- Write a one-page clinic protocol covering positioning, patient instructions, acceptable pain threshold for testing, and documentation fields.
- Define your “quality checks” for repeat tests (effusion, ROM, guarding notes) so trend interpretation is credible.
- Align measurement timepoints with triage decisions, prehab, post-op milestones, and return-to-sport windows, so the numbers change care rather than just fill charts.
Clinical references (PubMed)
1) 2024 – Nascimento et al. – DYNEELAX Robotic Arthrometer Reliability and Feasibility on Healthy and Anterior Cruciate Ligament Injured/Reconstructed Persons. – Transl Sports Med – DOI: 10.1155/2024/3413466 – PMID: 38654720 – PubMed
2) 2026 – Andre et al. – Comparable outcomes following combined ACL and ALL reconstruction using a 1-strand versus 2-strand back-and-forth technique: Propensity score matched study. – J Exp Orthop – DOI: 10.1002/jeo2.70658 – PMID: 41799400 – PubMed
3) 2024 – Unal et al. – GNRB® Knee Arthrometer: Inter- and Intra-observer Reliability and Learning Curve. – Cureus – DOI: 10.7759/cureus.70838 – PMID: 39493172 – PubMed
4) 2025 – Figueroa et al. – Relationship Between Quantitative MRI UTE T2* of ACL Autografts and BMI-Normalized Knee Laxity Within the First Year After ACL Reconstruction. – Am J Sports Med – DOI: 10.1177/03635465251368393 – PMID: 40970673 – PubMed
5) 2025 – Huang et al. – The Association Between Concomitant Meniscal Tear, Tibial Slope, Static Knee Position, and Anterior Knee Laxity in ACL-Deficient Patients. – Orthop J Sports Med – DOI: 10.1177/23259671251324186 – PMID: 40124192 – PubMed